Fibroin is the primary structural component of natural silk produced by the silkworm Bombyx mori and is one of the most extensively studied fibrous proteins in the field of natural biomaterials. Within the raw silk fiber, fibroin typically accounts for 70% to 80% of the total protein composition, while the remaining fraction mainly consists of sericin, an adhesive protein that coats and binds fibroin filaments together during cocoon formation.
From a biochemical perspective, fibroin belongs to the class of scleroproteins, which are structural proteins characterized by high stability, insolubility, and mechanical resistance. These characteristics allow silk to exhibit remarkable physical properties that combine tensile strength, elasticity, and lightness, making it one of the most high-performance biological materials found in nature.
Scientific interest in fibroin has grown significantly in recent years, particularly because of its biological properties and its versatility as a biomaterial. The combination of biocompatibility, biodegradability, and molecular self-assembly capability has made this protein the focus of extensive research in biotechnology, regenerative medicine, and advanced materials science.
Molecular structure
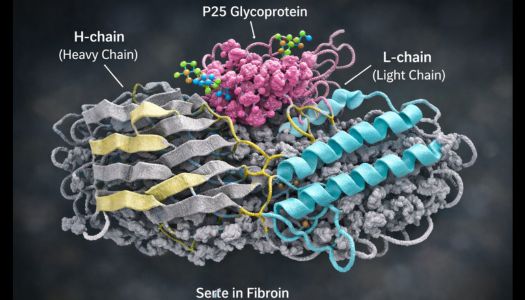
Silk fibroin possesses a highly organized molecular structure that plays a fundamental role in determining its mechanical and functional properties. The protein is mainly composed of a heavy chain (H-chain) and a light chain (L-chain) linked together through a disulfide bond. Associated with this complex is a glycoprotein known as P25, which contributes to the stabilization of the protein assembly during fiber formation.
The heavy chain represents the dominant component of the protein and has a molecular mass of approximately 350 kDa. From the standpoint of amino acid sequence, it is characterized by a highly repetitive structure dominated by glycine, alanine, and serine, amino acids that favor the formation of highly ordered structural domains.
These repetitive sequences frequently organize into motifs such as Gly–Ala–Gly–Ala–Gly–Ser, which promote the formation of antiparallel β-sheet secondary structures. Within the fiber, these structures pack tightly together, forming extremely compact crystalline domains.
Fibroin also exhibits a hierarchical architecture characterized by the alternation between highly ordered crystalline regions and more flexible amorphous regions. The crystalline domains provide significant mechanical strength, while the amorphous regions allow a certain degree of elasticity and deformation. This balance between rigidity and flexibility underlies the extraordinary mechanical properties of silk and represents one of the most sophisticated examples of structural engineering found in biological materials.
Physicochemical properties of silk fibroin
From a physicochemical standpoint, fibroin is intrinsically insoluble in most conventional solvents, including water, ethanol, and ether. However, it can be solubilized using specific solvent systems such as concentrated salt solutions or chaotropic agents, which allow the dissociation of secondary structures and enable the subsequent reprocessing of the protein.
One of the most relevant characteristics of fibroin is its amphiphilic nature, resulting from the alternation of hydrophobic and hydrophilic domains along the polypeptide chain. This property allows the protein to spontaneously self-assemble into stable supramolecular structures and to be transformed into a variety of material morphologies.
Through specific processing techniques, fibroin can be reorganized into numerous structural architectures, including thin films, hydrogels, nanofibers, microspheres, and three-dimensional scaffolds. These structures can be obtained through processes such as electrospinning, freeze-drying, phase separation, or molecular self-assembly, demonstrating the remarkable technological versatility of this protein.
From a biological standpoint, fibroin also exhibits a high degree of compatibility with living tissues. During certain processing procedures, bioactive peptide sequences may become exposed that promote cell adhesion through interactions with specific integrins located on the cell surface. This behavior facilitates the interaction between the biomaterial and biological systems, making fibroin particularly attractive for biomedical applications.
Another important aspect concerns the controlled biodegradability of the protein. Fibroin can be degraded by proteolytic enzymes present in the body, and the rate of degradation depends largely on the degree of crystallinity of the material. Structures with a higher crystalline organization tend to degrade more slowly, while more amorphous configurations are more easily resorbed.
Fibroin as an advanced biomaterial
The structural and biological properties of fibroin have led to its growing recognition as a reference biomaterial in several areas of scientific research. The combination of mechanical strength, structural stability, and biological compatibility makes this protein particularly suitable for the development of materials designed to interact with complex biological systems.
In the field of tissue engineering, fibroin is used to create three-dimensional scaffolds that mimic the architecture of the extracellular matrix. These structures act as supports for cell adhesion, proliferation, and differentiation, promoting the formation of new tissues.
Fibroin is also being investigated for the development of controlled drug delivery systems, in which the structure of the protein can be used to encapsulate therapeutic molecules and release them gradually over time. This approach allows modulation of the release kinetics of active compounds and can improve the effectiveness of pharmacological treatments.
Another area of application concerns materials for skin regeneration, where fibroin is studied for the production of advanced wound dressings and bioactive supports capable of promoting tissue repair processes. The protein’s ability to support cell proliferation and interact with the extracellular matrix plays a key role in these applications.
At the same time, advances in genetic engineering technologies are opening new perspectives in the development of modified fibroins containing specific bioactive sequences. Through recombinant DNA techniques, it is possible to incorporate functional peptides, growth factors, or antimicrobial molecules directly into the protein structure, further expanding the potential applications of this biomaterial.